Computational Psychiatry 2.0: Can We Simulate the Brain to Treat Schizophrenia Better?
 Introduction: Psychiatry Is Moving Beyond Symptom Checklists
Introduction: Psychiatry Is Moving Beyond Symptom Checklists
Psychiatry has always depended on careful listening.
A patient describes voices, fear, suspicion, withdrawal, lack of motivation, poor sleep, emotional blunting, cognitive slowing, or difficulty functioning. The psychiatrist listens, examines, understands the person’s context, makes a diagnosis, and begins treatment.
This clinical method remains irreplaceable.
But modern psychiatry is now entering a new phase.
We are slowly moving from asking only:
“What symptoms does this person have?”
to also asking:
“What brain circuits, neurotransmitter systems, and information-processing mechanisms may be producing these symptoms?”
This is where Computational Psychiatry 2.0 becomes important.
It is not about replacing the psychiatrist with a machine. It is about giving psychiatrists better tools to understand the brain, predict treatment response, reduce trial-and-error prescribing, and design more precise combinations of medication, psychotherapy, and neuromodulation.
A recent IUPHAR review by Hugo Geerts describes this emerging approach as a way to combine computational neuroscience, psychopharmacology, neuroimaging, neuromodulation, and clinical trial data into a unified model for schizophrenia treatment.
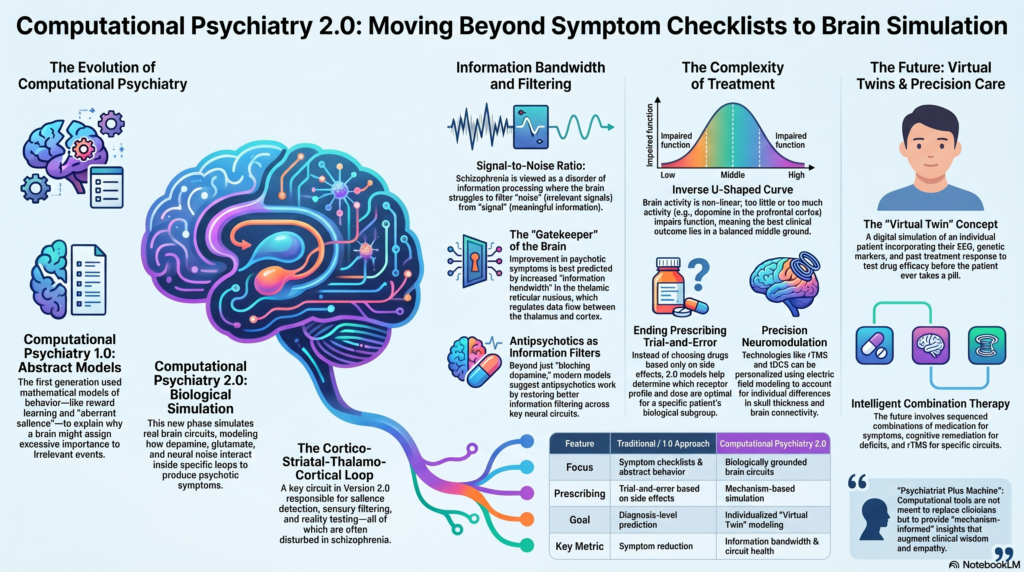
What Was Computational Psychiatry 1.0?
The first generation of computational psychiatry tried to explain mental illness using mathematical models of behaviour.
For example, in schizophrenia, researchers focused on dopamine, reward learning, prediction error, and salience.
Dopamine helps the brain decide what is important.
When dopamine signalling becomes dysregulated, the brain may start assigning excessive importance to neutral or irrelevant events. A passing glance, a random sound, or an ordinary coincidence may begin to feel deeply meaningful. This can contribute to suspiciousness, delusional interpretations, and abnormal salience.
These models helped explain psychosis at a deeper level.
They moved psychiatry beyond surface description and gave us concepts such as:
- Prediction error
- Aberrant salience
- Reinforcement learning
- Reward sensitivity
- Decision-making under uncertainty
- Impaired source monitoring
But there was a limitation.
These models often remained abstract. They could say that a person’s prediction error system was abnormal, but they could not always explain which brain circuit, which receptor, which neurotransmitter system, or which drug target was responsible.
That is where Computational Psychiatry 2.0 becomes different.
What Is Computational Psychiatry 2.0?
Computational Psychiatry 2.0 is a more biologically grounded version of computational psychiatry.
It tries to simulate real brain systems rather than only abstract psychological processes.
In schizophrenia, this means modelling key brain circuits such as the cortico-striatal-thalamo-cortical loop.
This circuit connects the cortex, striatum, thalamus, and back to the cortex. It is involved in:
- Salience detection
- Sensory filtering
- Attention
- Action selection
- Cognitive control
- Reality testing
- Behavioural regulation
These are precisely the functions that may become disturbed in schizophrenia.
Instead of saying “schizophrenia is due to dopamine imbalance,” Computational Psychiatry 2.0 asks a more precise question:
How do dopamine, glutamate, acetylcholine, serotonin, cortical activity, thalamic gating, and neural noise interact inside specific brain circuits to produce psychotic symptoms?
This is a major shift.
It moves psychiatry from diagnosis-level prediction to mechanism-level simulation.
Schizophrenia as a Disorder of Information Processing
Schizophrenia is not simply a disorder of hallucinations and delusions.
It is also a disorder of how the brain filters, organizes, and interprets information.
The brain is constantly flooded with signals: sounds, sights, memories, bodily sensations, emotions, thoughts, and social cues. A healthy brain must decide what is relevant and what can be ignored.
In schizophrenia, this filtering process may become disturbed.
Internally generated experiences may be misattributed to the outside world. Irrelevant events may feel significant. Ordinary perceptions may become emotionally charged. Ambiguous situations may be interpreted as threatening or personally meaningful.
This is often described as a problem in the brain’s signal-to-noise ratio.
The signal is meaningful information.
The noise is irrelevant or random activity.
When the signal-to-noise ratio becomes poor, the brain may struggle to separate reality from internally generated experience.
This is why the concept of information bandwidth becomes so interesting.
Information Bandwidth: A New Way to Understand Psychosis
One of the most important ideas discussed in the review is information bandwidth.
In simple terms, information bandwidth refers to the capacity of a brain circuit to transmit meaningful information.
A circuit with good information bandwidth can process signals efficiently.
A circuit with poor information bandwidth may either lose important information or allow too much noise to enter the system.
In the computational model described in the review, improvement in psychotic symptoms after antipsychotic treatment was best predicted by increased information bandwidth in the thalamic reticular nucleus.
This is clinically fascinating.
The thalamic reticular nucleus acts like a gatekeeper. It helps regulate the flow of information between the thalamus and cortex. If this gatekeeping system becomes disturbed, the brain may fail to filter sensory, emotional, and cognitive information properly.
This gives us a more refined way to understand antipsychotic response.
Antipsychotics may not only be “blocking dopamine.”
They may be helping restore better information filtering across key brain circuits.
Why More Brain Activity Is Not Always Better
A common misunderstanding is that more brain activity means better brain function.
That is not true.
The brain does not work like a simple engine where more firing means more performance.
In fact, too much firing can be chaotic. Too little firing can be inefficient. What matters is the timing, coordination, variability, and meaningfulness of neuronal activity.
This is where concepts such as entropy and inter-spike interval variability become relevant.
The brain must maintain a balance between order and flexibility.
Too much rigidity can impair adaptation.
Too much randomness can impair clarity.
In psychosis, the problem may not be only “excess dopamine” or “reduced glutamate.” It may involve a deeper disturbance in the way neural circuits process uncertainty, salience, and information.
Computational Psychiatry 2.0 gives us tools to study these patterns more precisely.
Why Psychiatric Treatments Are Not Linear
In medicine, we often like simple explanations.
More drug equals more effect.
More receptor blockade equals more symptom reduction.
More stimulation equals more improvement.
But the brain does not behave in such a simple linear manner.
Many psychiatric mechanisms follow an inverse U-shaped curve.
This means that too little activity is bad, too much activity is also bad, and the best clinical outcome lies somewhere in the middle.
Dopamine and prefrontal cognition are classic examples.
Too little dopamine can impair working memory and attention.
Too much dopamine can also impair working memory and attention.
The same may apply to glutamate modulation, negative symptoms, and cognitive enhancement.
This is why some drugs look promising in early trials but fail in later studies. The dose may be wrong. The patient subgroup may be wrong. The target may help one symptom domain while worsening another. The background antipsychotic may interfere with the new drug.
Computational Psychiatry 2.0 can help us model these complex non-linear interactions.
It allows us to ask:
- Which dose is likely to help?
- Which receptor profile is optimal?
- Which symptom domain may improve?
- Which domain may worsen?
- Which patient subgroup is most likely to benefit?
- What happens when this drug is added to an existing antipsychotic?
This is a more intelligent way to approach psychiatric treatment development.
From Antipsychotic Trial-and-Error to Mechanism-Based Treatment
Current antipsychotic prescribing still involves a degree of trial and error.
A clinician chooses a medication based on symptom profile, side-effect risk, previous response, cost, availability, adherence, metabolic risk, prolactin risk, sedation, extrapyramidal risk, and patient preference.
This is good clinical practice.
But it is still limited.
Two patients with schizophrenia may look similar clinically but differ biologically.
One may have prominent striatal dopamine dysregulation.
Another may have severe cortical hypofunction.
Another may have cognitive impairment worsened by anticholinergic burden.
Another may have negative symptoms that are primary, while another may have secondary negative symptoms due to depression, sedation, extrapyramidal symptoms, or social deprivation.
Computational models may eventually help separate these patterns.
Instead of asking only:
“Which antipsychotic works for schizophrenia?”
we may ask:
“Which receptor action, in which circuit, for which symptom domain, in which patient, at what dose, and in what combination?”
That is the promise of precision psychiatry.
Virtual Twins: A Future Direction
Another exciting idea is the concept of virtual twins.
A virtual twin is a digital simulation of an individual patient.
In the future, such a model may include:
- Clinical symptoms
- Cognitive profile
- EEG or QEEG findings
- Neuroimaging data
- Current medications
- Smoking status
- Drug metabolism variables
- Genetic markers
- Past treatment response
- Side-effect vulnerability
- Functional impairment
The model could then simulate how the patient may respond to different treatments.
For example:
Would this patient respond better to a D2 antagonist or a partial dopamine agonist?
Would adding a cholinergic agent improve cognition or worsen side effects?
Would rTMS help a specific symptom domain?
Would a medication increase plasticity during neuromodulation?
Would this patient be vulnerable to cognitive dulling from anticholinergic burden?
This is not yet routine clinical practice.
But it represents the direction psychiatry is moving toward.
Neuromodulation and Computational Psychiatry
Computational Psychiatry 2.0 is not limited to medication.
It is also highly relevant to neuromodulation.
Treatments such as rTMS, tDCS, ECT, magnetic seizure therapy, and neurofeedback can be understood as ways of perturbing brain networks.
But neuromodulation has a major challenge: individual variability.
The same rTMS protocol may produce different effects in different people because of differences in skull thickness, cortical anatomy, brain connectivity, coil placement, medication status, and illness biology.
This is where electric field modelling becomes useful.
By modelling how stimulation enters the brain, we may be able to personalize neuromodulation protocols.
In the future, we may combine:
- Structural MRI
- Functional MRI
- EEG/QEEG
- Electric field modelling
- Medication modelling
- Symptom-domain mapping
This could help clinicians decide not only where to stimulate, but also when, how intensely, and in combination with which medication or psychological intervention.
Combining Medication, Psychotherapy, and Neuromodulation
The future of schizophrenia treatment may not be one single drug.
It may be intelligent combination therapy.
A patient may require:
- Antipsychotic medication for positive symptoms
- Cognitive remediation for cognitive deficits
- Psychosocial rehabilitation for functioning
- Family intervention for relapse prevention
- rTMS or tDCS for specific circuits
- Digital monitoring for early warning signs
- Sleep and lifestyle correction
- Substance-use intervention when needed
- Structured psychotherapy for coping, insight, and recovery
The key question is not whether we should combine treatments.
The key question is:
Which combination, for which patient, at what stage, in what sequence, and with what biological target?
Computational Psychiatry 2.0 may help answer this.
It can allow researchers to simulate different combinations before testing them in large trials.
This may reduce failed trials, improve patient selection, and make psychiatric treatment more rational.
Why This Matters in Indian Psychiatry
In India, psychiatry faces unique challenges.
We see patients at different stages of illness. Many present late. Some have already received multiple medications. Adherence may be inconsistent. Family involvement is high. Cost matters. Stigma delays care. Substance use, sleep deprivation, stress, and medical comorbidities often complicate the picture.
In such a context, precision psychiatry cannot simply mean expensive genetic testing or imported technology.
It must mean clinically useful precision.
That includes:
- Better symptom-domain assessment
- Careful cognitive evaluation
- Rational antipsychotic selection
- Avoidance of unnecessary polypharmacy
- Monitoring metabolic and neurological side effects
- Using EEG/QEEG and cognitive tools where appropriate
- Integrating neuromodulation responsibly
- Personalizing psychoeducation and rehabilitation
Computational Psychiatry 2.0 offers a future framework for this.
It can help us move from “one-size-fits-all psychiatry” to “mechanism-informed psychiatry.”
A Word of Caution
Computational Psychiatry 2.0 is promising, but it should not be oversold.
These models are only as good as the data and assumptions that go into them.
Psychiatric symptoms are not just brain signals. They are also shaped by biography, trauma, culture, relationships, meaning, poverty, stigma, family systems, and social context.
A mathematical model cannot replace clinical understanding.
It cannot replace empathy.
It cannot replace the therapeutic relationship.
The best future is not machine instead of psychiatrist.
The best future is psychiatrist plus machine.
Clinical wisdom, careful listening, neuroscience, digital tools, computational modelling, and humane care must come together.
Conclusion: The Future Is Mechanism-Informed Psychiatry
Computational Psychiatry 2.0 represents a major step forward.
The first generation of computational psychiatry helped us understand mental illness through abstract models of learning, reward, prediction, and decision-making.
The second generation goes deeper.
It tries to connect symptoms with brain circuits, receptor pharmacology, drug exposure, neuroimaging, EEG, entropy, information bandwidth, and neuromodulation.
For schizophrenia, this is especially important.
Schizophrenia is not merely a disorder of dopamine. It is a disorder of distributed brain networks, impaired salience, altered signal-to-noise ratio, cognitive dysfunction, emotional and motivational disturbance, and real-world disability.
Computational Psychiatry 2.0 may help us design better drugs, improve clinical trials, personalize neuromodulation, reduce unnecessary polypharmacy, and eventually predict treatment response more accurately.
The future of psychiatry will not be less human because it becomes more scientific.
Done well, it can become more precise, more thoughtful, and more compassionate.
About the Author
Dr. Srinivas Rajkumar T, MD (AIIMS, New Delhi), DNB, MBA (BITS Pilani)
Consultant Psychiatrist & Neurofeedback Specialist
Mind & Memory Clinic, Apollo Clinic Velachery Opp. Phoenix Mall
✉ srinivasaiims@gmail.com 📞 +91-8595155808